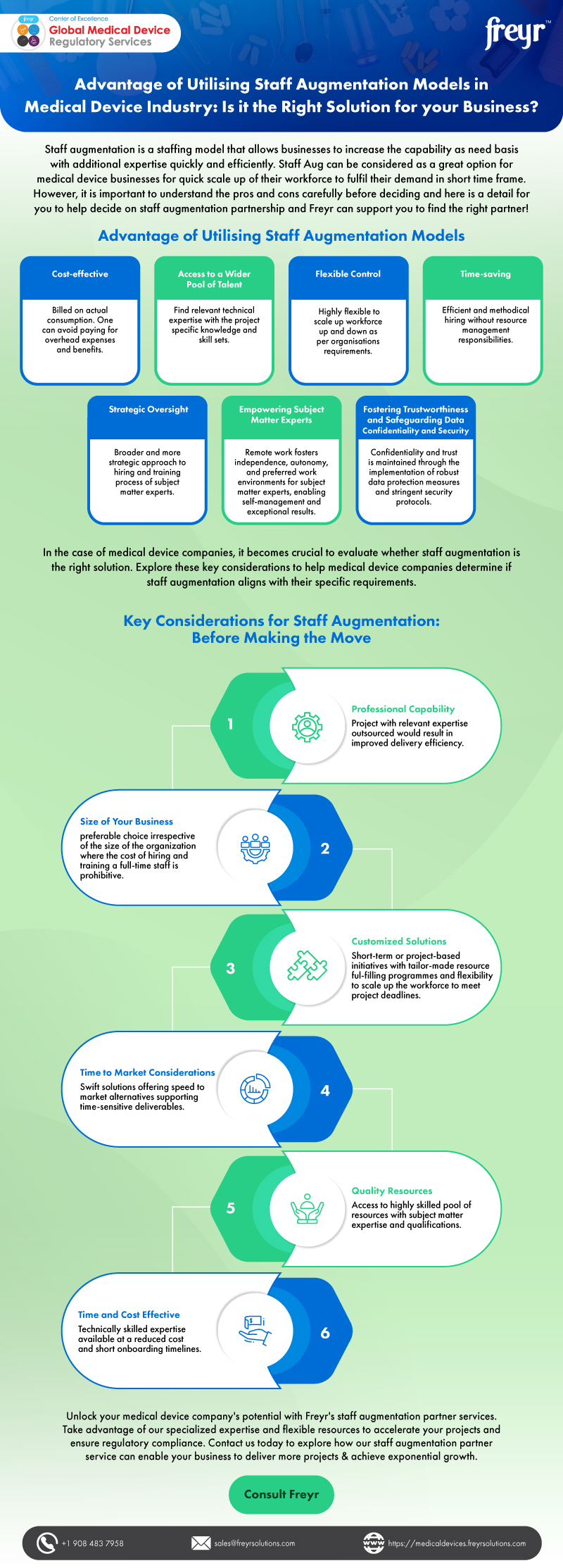
Freyr provides a very reliable service with expertise across many countries. I can rely upon Freyr to provide the information necessary to make an informed decision before entering a formal scope of work agreement. Once a project is underway, the Freyr team act professionally to execute the work with excellent communication of progress.
-Regulatory and Quality Assurance Manager,
Numedico Technologies